LEVEL 5, Senior Assistant Chemist.
THEORY
1. Acids and bases: Bronsted and Lewis acid-base concept, hard and soft acids and
bases, relative strengths of acids and bases and effect of substituents and solvents on
them.
2. Refining and purification of metals: Chromatography, ion exchange, solvent
extraction, oxidative refining, parting process, zone refining, Mond's process.
3. Liquid state: Vapour pressure, vapour pressure and boiling point, surface tension and
its determination using Stalagnometer, viscosity and determination by Ostwald
viscometer, applications of surface tension and viscosity measurements, flash point.
4. Solid state: Crystalline and amorphous solids, classification of solids on the basis of
dominant type of bond.
5. Chemical Kinetics: Concept of rate of reaction, dependence of reaction rate on
concentration, measurement of reaction rate, order and molecularity of a reaction,
rate equations for zero, first and second order reactions, the temperature dependence
of reaction rates, reaction mechanisms, catalysis 2
6. Chemical change and chemical equations: Type of chemical change, factors
governing chemical change, chemical equations, their significance different ways of
balancing them.
7. Hydrogen: Position of hydrogen in the periodic table, technical preparation,
Laboratory preparation from acids.
8. Oxygen: Position in the periodic table, laboratory preparation from compound
containing high percentage of oxygen, properties, applications, oxides.
9. Water: Hard and soft water, removal of hardness, composition of water, structure
solvent property of water.
10. Carbon Mono-oxide and Carbon Dioxide: Preparation, properties and applications
11. Sulphur: Structure of sulphur
12. Hydrogen sulphide and sulphur dioxide: Preparation properties and applications
13. Sulphuric Acid: Properties and applications
14. Phosphorus: Know about red and white phosphorus
15. Isolation and purification of organic compounds: Sources of compound,
characteristics of organic compound, Extra action of organic substances form natural
sources, purification, crystallization, sublimation, distillation, steam distillation,
washing drying, criteria of purify.
16. Aliphatic and aromatic Hydrocarbons and petro chemicals.
17. Antiknocking properties of petro chemicals.
PRACTICAL
The candidates will be asked to perform laboratory works from the following topics.
1. Determination of volume, weight etc using deferent measuring tools
2. Determination of Density of materials
3. Determination of refractive index
4. Determination of specific heat capacity and latent of materials
5. Determination of specific gravity
6. To separate deferent materials from the mixture.
7. Use of distillation to produce distillate
8. To neutralize acid with proper solution.
9. To investigate the composition of water by electrolysis.
10.To determine the pH of different unknown solution & using pH paper and universal
indicator.
11.To carry out conductivity experiments on solids & liquids
12.Determine Flash point, Viscosity of Oil
LEVEL 4, Senior Assistant Chemist.
1. Periodic classification of elements and physical properties
Periodicity of elements, s,p,d and f blocks, long form of periodic table, discussion on
properties like atomic, ionic and covalent radii, ionization potential, screening or
shielding effect, electronegetivity, electron affinity.
2. Acids and bases
Bronsted and Lewis acid- base concept, hard and soft acids and bases, relative strengths
of acids and bases and effect of substituents and solvents on them, pH and H
+
ion
concentration of strong acids, neutralization.
3. Principles of qualitative and quantitative analysis
Solubility product, common ion effect, their application in group separation, principles
of volumetric and gravimetric analysis.
4. Environmental pollution
An elementary study of environmental pollution (in air and water) arising due to the
presence of dust, carbon, CO, CO2, NOx, Sox, H2S and heavy metals.
5. Refining and purification
Chromatography (column, paper), ion exchange, solvent extraction, oxidative refining,
parting process, zone refining, fractional distillation.
6. Carbon
Allotropes of carbon, preparation and properties of oxides of carbon (CO, CO2), general
knowledge of source CO2 emission in atmosphere, photosynthesis and carbon
sequestration.
7. Water
Structure of water, solvent properties of water, hard and soft water, hydrogen bonding.
1. Organic reaction mechanism
Energetic of reaction, energy profile diagrams, exothermic and endothermic reactions,
types of mechanisms,thermodynamic and kinetic requirement of reaction, Methods of
determining mechanism.
2. Substitution and elimination reactions
Structure of alkyl halides, nucleophilic aliphatic substitution reactions, nucleophiles and
leaving groups, the SN2 reaction (kinetics, mechanism, stereo chemistry and reactivity),
The SN1 reaction (kinetics, mechanism, stereo chemistry and reactivity), structure of
alkenes, the E2 reaction (kinetics, mechanism, orientation and reactivity), the E1
reaction (kinetics, mechanism, orientation and reactivity).
3. Aromaticity
Aliphatic and aromatic compounds, structure of benzene, Kekule structure, stability of
benzene ring, Huckel's 4n+2 rule, electropilic aromatic substitution reaction, effect of
substituent group, orientation, mechanism of electrophilic aromatic substitution reaction.
4. Spectroscopic techniques
An elementary study of organic compounds structural elucidation by uv, ir, nmr and
mass techniques.
5. Purification of organic compounds
Methods of purification of crude organic compound, determination of purity of organic
compounds, principles and practices behind identification of functional group in organic
compounds.
6. Hydrocarbons
Classification of hydrocarbons, sources of hydrocarbons, nomenclature. Aliphatic and
aromatic hydrocarbons. Alkanes, alkenes and alkynes; nomenclature, preparation,
properties and uses. General knowledge of petrochemical compounds, fossil fuels, flash
point, auto ignition temperature, octane number, anti-knocking .7. Alcohols
Classification, distinction between 1°, 2° and 3° alcohols, industrial preparation of
ethanol, uses of ethanol in industries and as vehicle fuel.
8. Carbohydrates
Classification of carbohydrates, structure of glucose, chemical conversion to alcohol.
1. Atomic structure
Sub atomic particles, their masses and charges, atomic mass unit, Daltons atomic theory,
Rutherford's experiment, Bohr's model, Heisenberg uncertainty principle, Paulis
exclusion principle, Hunds rule of maximum multiplicity, elementary idea of quantum
mechanical model of atom, quantum numbers, isotopes and fractional weights, nuclear
fission and fusion.
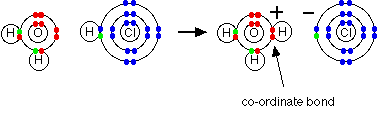
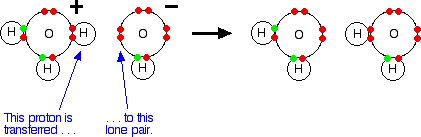
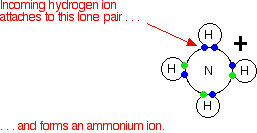
2. Chemical bonding
Valency, octate rule, chemical bonds and lewis structure, ionic and covalent bonds, ionic
character of covalent bond, coordinate covalent bond, metallic bonds, intermolecular and
Van der Wall's forces, shapes of compound such as NH3, CH4, PF5, BF3,
3. Oxidation and Reduction
Electronic concept of oxidation and reduction reactions, oxidation number, balancing
redox reaction by oxidation number and ion electron methods.
4. Chemical equilibrium
Equilibrium in physical processes, features of dynamic equilibrium, LeChatelier's
principle: effect of pressure, temperature, concentration and catalyst on chemical
equilibrium, equilibrium constant Kp and Kc
5. Gaseous State
Postulates of kinetic molecular theory and their significance, Boyle's law, temperature
scale and Charle's law, Dalton's law of partial pressure the ideal gas law, Molecular
diffusion and Graham's law, molecular collision and mean free path, intermolecular
forces, critical temperature and pressure, departures from ideal gas law, Vander waals
constants a and b, liquefaction of gases.
6. Liquid state
Vapour pressure, vapour pressure and boiling point, surface tension and its determination
using Stalagnometer, viscosity and determination by Ostwald viscometer, applications of
surface tension and viscosity measurements.
7. Solid state
Crystalline and amorphous solids, classification of solids on the basis of dominant type
of bond.
8. Chemical Kinetics
Concept of rate of reaction, dependence of reaction rate on concentration, measurement
of reaction rate, order and molecularity of a reaction, rate equations for zero, first and
second order reactions, temperature dependence of reaction rates, reaction mechanisms,
catalysis
9. Thermodynamics
First law of thermodynamics, thermodynamic terms, iso thermal but not reversible
expansion of an ideal gas, iso thermal reversal expansion of an ideal gas, enthalpy of
physical and chemical changes, Hess's law of constant heat summation, spontaneous and
non-spontaneous changes, second law of thermodynamics, spontaneity and entropy
change, entropy as a measure of randomness, a molecular interpretation of entropy, the
free-energy function and equilibrium, free energy and temperature, free energy and the
equilibrium constant, criteria of spontaneity.
D) GENERAL1. Treatment of analytical data
Nature of analytical measurements, significant figures, precision and accuracy, errors,
basic statistical concepts, average and measures of dispersion, standard deviation,
confidence limits, elements of standards and measures. Data processing in spread sheets.
2. Laboratory management
General idea of safety precaution in the laboratory, care and maintenance of laboratory
equipments.